
by Vita | نوفمبر 26, 2017 | Articles, Articles2
Click to Go to FORM Click To help you find answers

by Vita | نوفمبر 8, 2016 | Articles
Carbonyl iron Pregnancy Warnings Carbonyl iron has not been formally assigned to a pregnancy category by the FDA. There are no controlled data in human pregnancy. Human case reports have not revealed evidence of teratogenicity. Carbonyl iron should only be given...

by Vita | أكتوبر 14, 2016 | Articles
drugs.com Those memory lapses many women notice around menopause are real, and they can begin at a relatively young age, researchers report. It’s common for women going through menopause to complain of what researchers sometimes call “brain fog”...

by Vita | أكتوبر 12, 2016 | Articles
Key to grades A Strong scientific evidence for this use B Good scientific evidence for this use C Unclear scientific evidence for this use D Fair scientific evidence against this use (it may not work) F Strong scientific evidence...
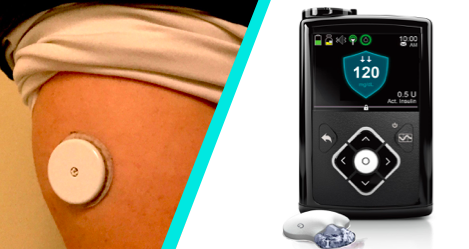
by Vita | سبتمبر 30, 2016 | Articles
The first automated insulin delivery device for type 1 diabetes has been approved by the U.S. Food and Drug Administration for people aged 14 and older. Often called an “artificial pancreas,” the MiniMed 670G hybrid closed loop system automatically...
by Vita | سبتمبر 7, 2016 | Articles
Taking antibiotics at a very young age could increase the risk of certain allergies later in life, new research suggests. “Early life exposure to antibiotics is related to an increased risk of both eczemaand hay fever later in life,” said Fariba Ahmadizar...







